Lipedema Common Case Report Form (CCRF)
Advancing Lipedema Research Through Standardization
What is the Lipedema CCRF?
The Lipedema Common Case Report Form (CCRF) is an important step in strengthening the research infrastructure for Lipedema.
Developed through international collaboration and broad stakeholder engagement, the Lipedema CCRF is a unified, publicly available framework designed to improve data quality, reproducibility, and comparability across studies. By supporting more consistent data collection, it helps researchers generate findings that can be meaningfully compared and evaluated. To learn more about the development process and the consensus methodology behind the CCRF, read the full paper here.
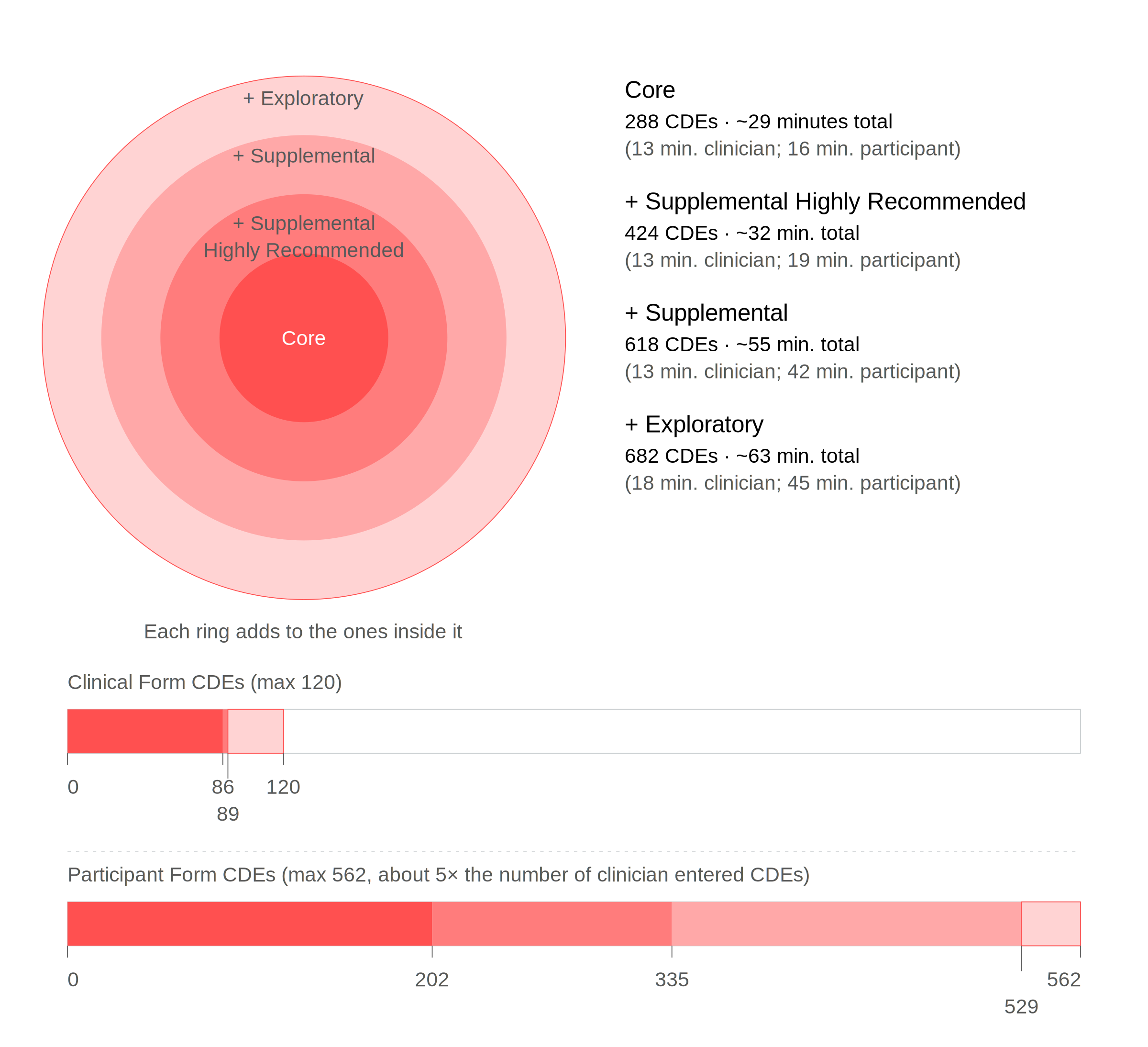
The Lipedema CCRF is organized into Exploratory, Supplemental, Supplemental Highly Recommended, and Core classifications. While the Foundation recommends researchers use the most inclusive version their study design allows, the classification structure allows for standardization of essential data elements (standardized questions with defined response options) while maintaining flexibility for different research designs and clinical settings.
The Lipedema CCRF helps researchers:
Assess the applicability of findings from individual studies
Compare results across studies
Conduct rigorous meta-analyses
Conduct retrospective analyses to inform diagnosis standardization
Identify and classify meaningful subtypes
As adoption grows, the Lipedema CCRF will support stronger meta-analyses, more consistent diagnostic research, and a stronger evidence base for treatment, policy, and patient care.
How to use the Lipedema CCRF
Step 1: Download and share the Lipedema CCRF with PI(s), study clinicians, and other key stakeholders to determine which set of questions to use (the Foundation strongly recommends use of the longest form feasible).
Step 2: Complete an Intention of Use Form to let us know how you plan to use the Lipedema CCRF.
Step 3: Download the appropriate data dictionary.
Step 4: Collect data.
Step 5: Post-study, send any shareable data to the Foundation at biobank@lipedema.org.
All CCRF versions are available in this download, organized across separate tabs. The Exploratory version is the complete CCRF, and researchers are encouraged to use the most comprehensive version their study design allows.
| CDE Classification | Definition | Number of CDEs |
|---|---|---|
| Exploratory | Core + Supplemental Highly Recommended + Supplemental + Data elements that are emerging, experimental, or do not yet have a clear link to Lipedema. These elements may represent novel assessments, biomarkers, or patient-reported outcomes not yet validated with Lipedema populations. Exploratory items can inform hypothesis generation and future standardization but are not yet recommended for routine use. |
Total CDEs: 682
|
| Supplemental | Core + Supplemental Highly Recommended + Data elements that may add scientific or clinical value but are not critical to diagnosis or characterization in all studies. These are useful in specific research aims or contexts, such as exploring specifics (such as triggers) for disease onset or worsening, treatment outcomes, quality of life, or comorbid conditions. |
Total CDEs: 618
|
| Supplemental Highly Recommended |
Core + Data elements that are not required for a basic diagnosis of Lipedema but are strongly recommended for comprehensive characterization of the condition and/or samples. These elements provide important context about subpopulations/subtypes and foundational understanding of disease onset, worsening (progression or exacerbation), severity, or impact and are widely applicable across most Lipedema studies. |
Total CDEs: 424
|
| Core | Common data elements related to the diagnosis of Lipedema include items essential for identifying, confirming, or differentiating Lipedema, as well as those providing clinically relevant information for diagnosis. Core elements include clinically assessed items such as Lipedema diagnosis for research purposes, Lipedema stage, anthropometric measurements, presence of edema, and palpable findings of nodularity or fibrosis. Participant-reported items that support diagnosis are also included, such as pain or discomfort in affected areas, common comorbidities, current medications and supplements, surgical history, menstrual status, family history, and basic demographics. This classification addresses areas of clinical disagreement and research gaps, facilitating data collection for cross-study and cross-geography comparisons to enhance understanding. |
Total CDEs: 288
|
Lipedema CCRF Data Dictionaries
The data dictionary versions of the Lipedema CCRF that define each question on the form: what it means, how responses should be recorded, and how the data is structured behind the scenes. Data dictionaries serve as a “rulebook” to ensure every question is interpreted consistently and the data can be used reliably.
Intention of Use Form
If you plan to use the Lipedema CCRF, please submit the information below.